Hill Analysis
of Interactive Ligand Binding (lig)
In this exercise, the ligand binding interactions between the bivalent
ligand binding sites of symmetrical dicarboxylic acid are examined using
a Hill plot. When fully
protonated ("saturated"),
a dicarboxylic acid undergoes a two-step
dissociation reaction with decreasing hydronium ion concentration.
When completely deprotonated ("desaturated"), a dicarboxylate undergoes a two-step
association reaction with increasing hydronium ion concentration.
For correct statistical analysis is it necessary to keep
track of binding to the right and left sides of the molecule.
| Two-step dissociation |
| | kd1 |  |  | | HOOC-CH2-COOH |
- H+ |  +H+ +H+ |
| -OOC-CH2-COOH |  | | kd1' | | | ka2 |  | | kd2 |
- H+
 | 
+ H+ |
| 
ka2' | | 
ka1 |

+ H+ | - H+
 |
| |  | ka1' | | |  | HOOC-CH2-COO- |
 +H+ +H+ | - H+  |
| -OOC-CH2-COO- |  |  |  | kd2' | | |
|
| Two-step association |
| kd1-cooh = [H+] * [-OOC-CH2-COOH] / [HOOC-CH2-COOH] (step d1) | | kd1'-cooh = [H+] * [HOOC-CH2-COO-] / [HOOC-CH2-COOH] (step d1') | | kd2-cooh = [H+] * [-OOC-CH2-COO-] / [-OOC-CH2-COOH] (step d2) | | kd2'-cooh = [H+] * [-OOC-CH2-COO-] / [HOOC-CH2-COO-] (step d2') | | kd1 = kd1' = 1/ka2 = 1/ka2' | kd2 = kd2' =1/ka1 = 1/ka1' |
| | ka1-cooh = [-OOC-CH2-COOH] / [ H+] * [-OOC-CH2-COO-] (step a1) | | ka1'-cooh =[HOOC-CH2-COO-] / [H+] * [-OOC-CH2-COO-] (step a1') | | ka2-cooh = [HOOC-CH2-COOH] / [H+] * [-OOC-CH2-COOH] (step a2) | | ka2'-cooh = [HOOC-CH2-COOH] / [H+] * [HOOC-CH2-COO-] (step a2') | | ka1 = ka1' = 1/kd2 = 1/kd2' | ka2 = ka2' = 1/kd1 = 1/kd1' |
|
Continue the acid-base titration analysis of dicarboxylic acid by
opening the Excel spreadsheet below.
- Up and down arrow dials adjust the pH1/2, i.e., the midpoint, and the slope of a straight line.
Dicarboxylic Acid Titration:
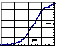
Hill Plot
© Duane W. Sears
Revised: August 10, 1998
![]()